The Next Generation for Canine Glaucoma. Triple Therapy in One Step.
The Next Generation for
Canine Glaucoma.
Triple Therapy in One Step.
The Next Generation for Canine Glaucoma.
Triple Therapy in One Step.
This medical CE program provides veterinarians and veterinary technicians with advanced clinical training in emerging medical therapy for canine glaucoma and dry eye disease (DED/KCS). The course explores the interaction between intraocular pressure dynamics, ocular surface inflammation, and tear film integrity, with specific emphasis on the role of novel lipid-base ocular surface formulations used in conjunction with advanced tear replacers to promote long-term corneal health and visual function.
Glaucoma is a group of ocular disorders characterized by elevated intraocular pressure (IOP) that can progressively impair vision if unmanaged. Medical therapy to reduce IOP is recommended to delay onset and slow disease progression. However, many canine patients require multiple ocular hypotensive medications to achieve and maintain physiological IOP levels.
Stokes Healthcare is proud to work with Santgar, a leading veterinary pharmaceutical innovator in Mexico known for its research-driven, high-quality animal health solutions. Santgar sought us out for this collaboration, recognizing our product quality, rigorous processes, and shared commitment to the same standards and values. Together, we are introducing new ophthalmic formulations to the U.S. market, expanding access to advanced veterinary therapies and improving patient outcomes.

Clinical evidence in glaucoma patients consistently demonstrates that combination pressure-lowering therapies reduce IOP more than monotherapy, with comparable or improved tolerability profiles (Boyle et al., 1998; Fechtner & Realini, 2004).
Travosant Forte® combines three mechanisms of action into a single ophthalmic gel formulation:
This triple-combination therapy delivers robust IOP reduction, achieving 20–30% greater pressure lowering from baseline.
Recommended Dosing:
Instill one drop every 12 hours in affected eye(s) and every 24 hours at night in unaffected eye. (Can adjust dose to every 8 hours in affected eye if needed.)
Important: Treat BOTH eyes even when only one is affected to delay glaucoma onset in the second eye. Travosant Forte works best when administered at consistent times daily. Treatment should be continued on a long term basis in patients in which intraocular pressure tends to remain elevated.
This triple fixed-combination gel system helps minimize dilution or washout of active ingredients that can occur when multiple medications are administered in close succession.
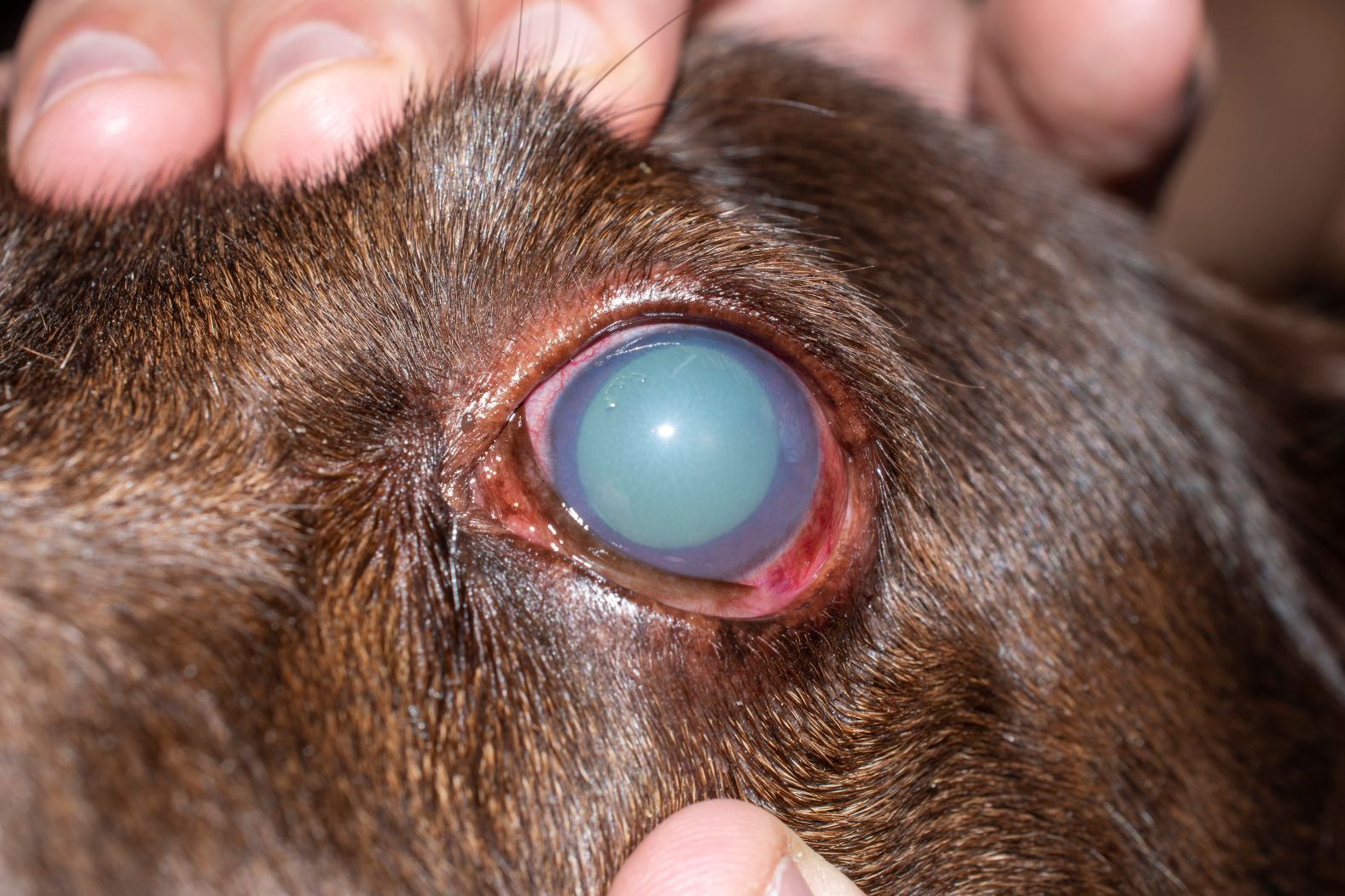

Yes. It is safe in uncertain cases and will not lower pressure in normal eyes.
Yes. Treating both eyes is critical to delaying disease in the second eye.
Travosant Forte™ works through a dual mechanism – it both reduces aqueous humor production and increases fluid drainage from the eye, lowering intraocular pressure (IOP).
What makes it unique is the inclusion of betaxolol, which not only reduces fluid production but also provides a neuroprotective effect. It penetrates to the optic nerve and retina, helping protect nerve cells and optic nerve fibers while pressure is being lowered – something not typically seen in standard glaucoma therapies.
In an acute setting, Travosant Forte™ can be administered every 1–2 hours until intraocular pressure (IOP) decreases to a safer range. Once pressure is controlled, dosing should transition to a maintenance schedule.
According to Dennis Brooks, DVM, PhD, DACVO, timolol may help reduce IOP but does not penetrate into the back of the eye and lacks neuroprotective effects. Betaxolol, however, penetrates into the retina and optic nerve and may provide neuroprotection by stabilizing calcium channels.
Once IOP has stabilized and the patient is clinically improved, dosing can typically be reduced to:
Travosant Forte™ is highly effective at rapidly lowering IOP and may be used as a primary therapy. However, clinical judgment should guide whether additional treatments are needed based on severity and patient response.
In many cases, intraocular pressure begins to decrease within 30 minutes to a few hours.
Yes. Pain relief occurs as pressure decreases – often rapidly.
Travosant Forte™ works best when administered at consistent times daily, which helps maintain stable intraocular pressure control.
Gustavo Adolfo García Sánchez, DVM, PhD, DACVO recommends administering the triple-drop medication three times daily in severely affected eyes and once nightly in the unaffected eye. If intraocular pressure improves, he reduces dosing in the affected eye to twice daily.
Eye pressure often increases at night. Evening dosing provides protection during this period.
According to Dennis Brooks, DVM, PhD, DACVO, it is theoretically possible, but unlikely. Gustavo Adolfo García Sánchez, DVM, PhD, DACVO added that in humans very low pressure can cause macular edema, but they have not observed retinal or macular edema in dogs using this treatment. If pressure drops too low, they suggested reducing frequency to every other day or a few times weekly.
Gustavo Adolfo García Sánchez, DVM, PhD, DACVO said he is happy when pressures reach 15–18 mmHg, and sometimes pressures as low as 8–9 mmHg are still well tolerated.
Yes, but you should wait at least 5 minutes after applying Travosant Forte™ before administering any other ophthalmic medications to ensure proper absorption.
No. Glaucoma requires lifelong management. Stopping treatment will result in pressure rising again.
No withdrawal effects, but untreated periods may allow further optic nerve damage.
Treatment should be continued on a long-term basis in patients where intraocular pressure tends to remain elevated, as glaucoma is a chronic condition requiring ongoing management.
Travosant Forte™ should be used with caution or avoided in certain cases, including:
Use should always be guided by a veterinarian, with monitoring as needed in at-risk patients.
Yes it can be used in both types of glaucoma. Gustavo Adolfo García Sánchez, DVM, PhD, DACVO suggested that it may perform particularly well in open-angle cases, especially when the lens remains positioned behind the iris.
Yes. Gustavo Adolfo García Sánchez , DVM, PhD, DACVO said even in blind eyes the medication can make patients significantly more comfortable and reduce redness and inflammation.
Most patients tolerate Travosant Forte™ well. The gel formulation is designed to be soothing. However, side effects could be burning, stinging or redness of the eye. Long term use of prostaglandins may cause darkening of the iris.
Yes. Travosant Forte™ should be refrigerated to maintain stability and effectiveness.
Travosant Forte™ is compounded in accordance with USP guidelines and is dispensed with the maximum allowable beyond-use date available from a compounding pharmacy. It will have a beyond-use date of approximately 7–30 days from the time of dispensing.
Yes. Travosant Forte™ is used in dogs, cats, and horses. It is most extensively studied in dogs, with additional clinical use showing effectiveness in horses and cats. Because glaucoma differs between species, treatment should always be guided by a veterinarian.
Caution is needed when using prostaglandin analogs in horses with glaucoma associated with uveitis. Uveitis is a leading cause of blindness in adult horses and can contribute to the development of Glaucoma, cataracts, and vascular damage within the eye. If the uveitis is active—indicated by signs such as aqueous flare (inflammation in the anterior chamber)—prostaglandin analogs are generally not recommended, as they may exacerbate inflammation. However, if the uveitis is inactive (no aqueous flare present), the inflammatory process is considered controlled. In these cases, using Travosant Forte™ may be appropriate as part of a glaucoma management plan. Because glaucoma in horses is complex and often multifactorial, treatment decisions should always be based on careful clinical evaluation and ongoing monitoring.
Gustavo Adolfo García Sánchez, DVM, PhD, DACVO noted that controlled studies have been conducted, and statistical analysis is currently ongoing. He added that publication has been delayed due to peer reviewers requesting additional and more extensive statistical evaluation.
The information provided in these FAQs is based on data and educational materials supplied by Santgar. This content is intended for educational and informational purposes only. While every effort has been made to ensure that the information presented is accurate and current at the time of publication, veterinary medicine is an evolving field, and new research may become available. The information on this site is not intended to replace the advice, diagnosis, or treatment recommendations of a licensed veterinarian. Veterinarians should use their professional judgment and consult relevant prescribing information and clinical resources when determining the most appropriate treatment for their patients. Pet owners should always consult their veterinarian regarding any questions about their pet’s health or treatment plan.
References:
Boyle, J. E., Ghosh, K., Gieser, D. K., & Adamsons, I. A. (1998). A randomized trial comparing the dorzolamide-timolol combination given twice daily to monotherapy with timolol and dorzolamide. Ophthalmology, 105(10), 1945–1951. https://doi.org/10.1016/s0161-6420(98)91046-6
Fechtner, R. D., & Realini, T. (2004). Fixed combinations of topical glaucoma medications. Current Opinion in Ophthalmology, 15(2), 132–135. https://doi.org/10.1097/00055735-200404000-00013